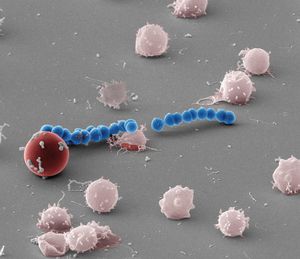
 Scanning electron micrograph of red blood cell hemolysis by the Streptolysin S producing Group A Streptococcus. Credit: Shaun Lee, Dustin Higashi
Scanning electron micrograph of red blood cell hemolysis by the Streptolysin S producing Group A Streptococcus. Credit: Shaun Lee, Dustin Higashi
One bacterial pathogen is responsible for a range of diseases, from pharyngitis and impetigo to more severe diagnoses such as toxic shock syndrome and necrotizing fasciitis (flesh eating disease), a serious bacterial skin infection that spreads quickly and kills the body’s soft tissue. The pathogen, known as Group A Streptococcus, remains a global health burden with an estimated 700 million cases reported annually, and more than half a million deaths due to severe infections.
The ability of Group A Streptococcus (GAS) to induce rapid destruction of red blood cells has been observed for more than a century and remains a clinical hallmark of GAS diagnosis. This destruction is due to the production of a small peptide toxin by GAS known as Streptolysin S (SLS).
Although it has been widely held that SLS exerts its lytic activity — the excessive destruction of red blood cells — through membrane disruption, its exact mode of action has remained unknown.
“Recent molecular studies by our lab and others have demonstrated that SLS is a peptide toxin linked to a broad class of bacterially produced compounds known as bacteriocins,” Shaun Lee, an associate professor of biological sciences at the University of Notre Dame, said. “Many of these related bacteriocins have defined cellular targets and have not been shown to function as general lytic agents of cellular membranes.”
In a new study, Lee’s research group provides the first real-time, high-resolution observation of Group A streptoccocal red cell destruction, also called beta-hemolysis.
“We demonstrate that the long-observed red blood cell hemolysis by SLS is not caused by general destruction of the red blood cell membrane, as has been previously thought, but rather that the action is due to the ability of the SLS toxin to directly target a specific outer membrane protein on the surface of the red blood cell, the major erythrocyte anion exchange protein Band 3.”
Importantly, chemical inhibition of Band 3 function completely blocked the hemolytic activity of SLS, and significantly altered the pathology induced by GAS in an in vivo skin infection model.
“Our studies provide the first mechanistic look into the longstanding question of SLS function and, importantly, open new therapeutic avenues for the treatment of severe GAS disease,” Lee said.
“This was a wonderful collaborative effort led by Dustin Higashi, a senior researcher in my lab, to try to answer the longstanding mystery of how this very powerful toxin known as Streptolysin S lyses red blood cells to contribute to invasive human disease caused by the Group A Streptococcus,” he said.
“Findings critical to the support of our hypothesis were provided by in vivo studies performed at the W.M. Keck Center for Transgene Research, under the direction of Francis Castellino and Victoria Ploplis. Using humanized mouse models, Keck scientists Deborah Donahue and Jeff Mayfield demonstrated that by blocking the action of SLS toxin during a GAS infection, the pathology at the site of the infection could be drastically reduced. These findings have tremendous potential for developing novel therapeutics to treat severe diseases caused by Group A Streptococcus.”
The study was supported by an NIH Innovator Grant awarded to Lee and appears in the journal Nature Microbiology. It can be found here: http://www.nature.com/nmicrobiol/.
Contact: Shaun Lee, 574-631-7197, lee.310@nd.edu
Originally published by William G. Gilroy at news.nd.edu on January 28, 2016.
